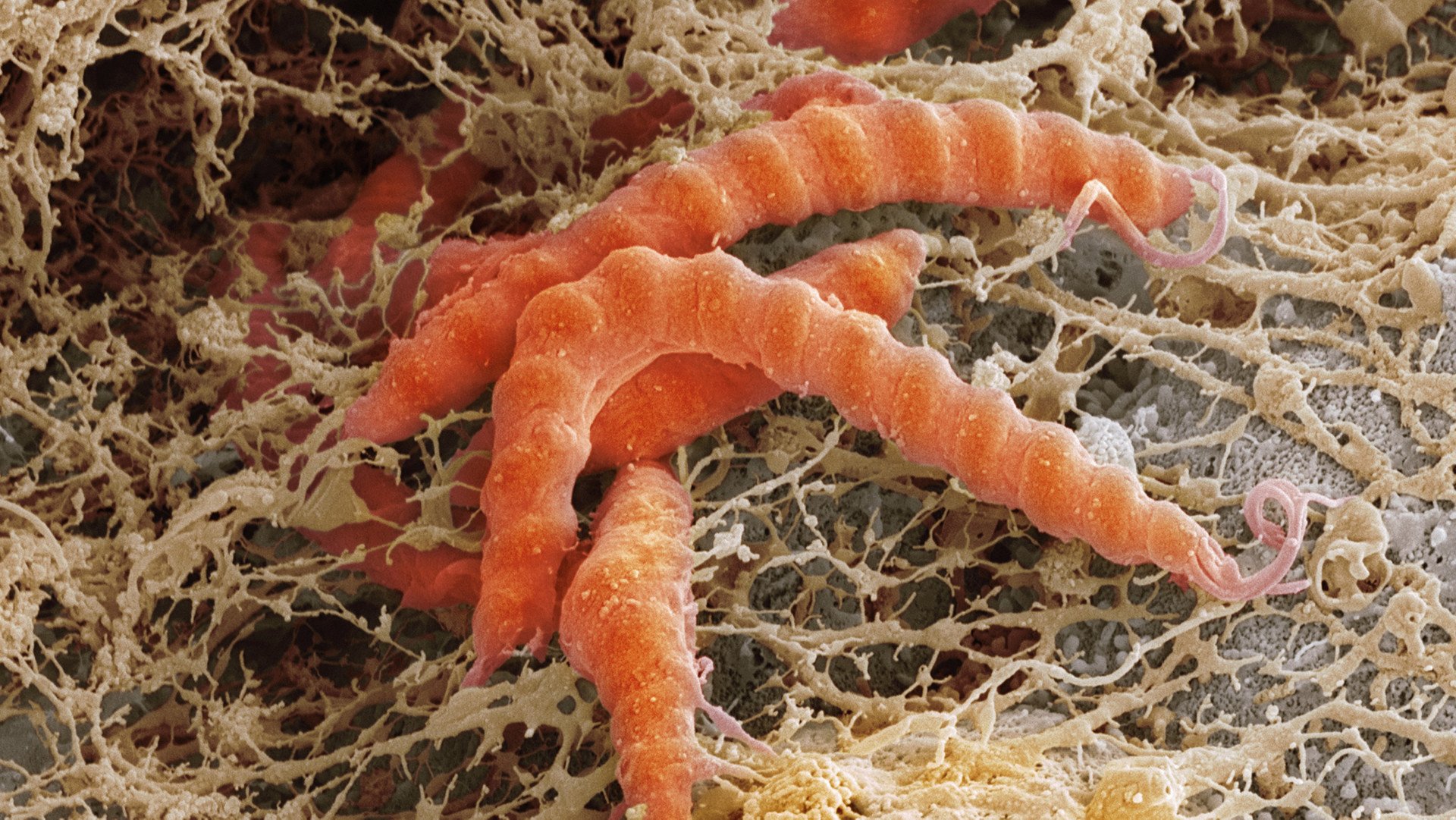
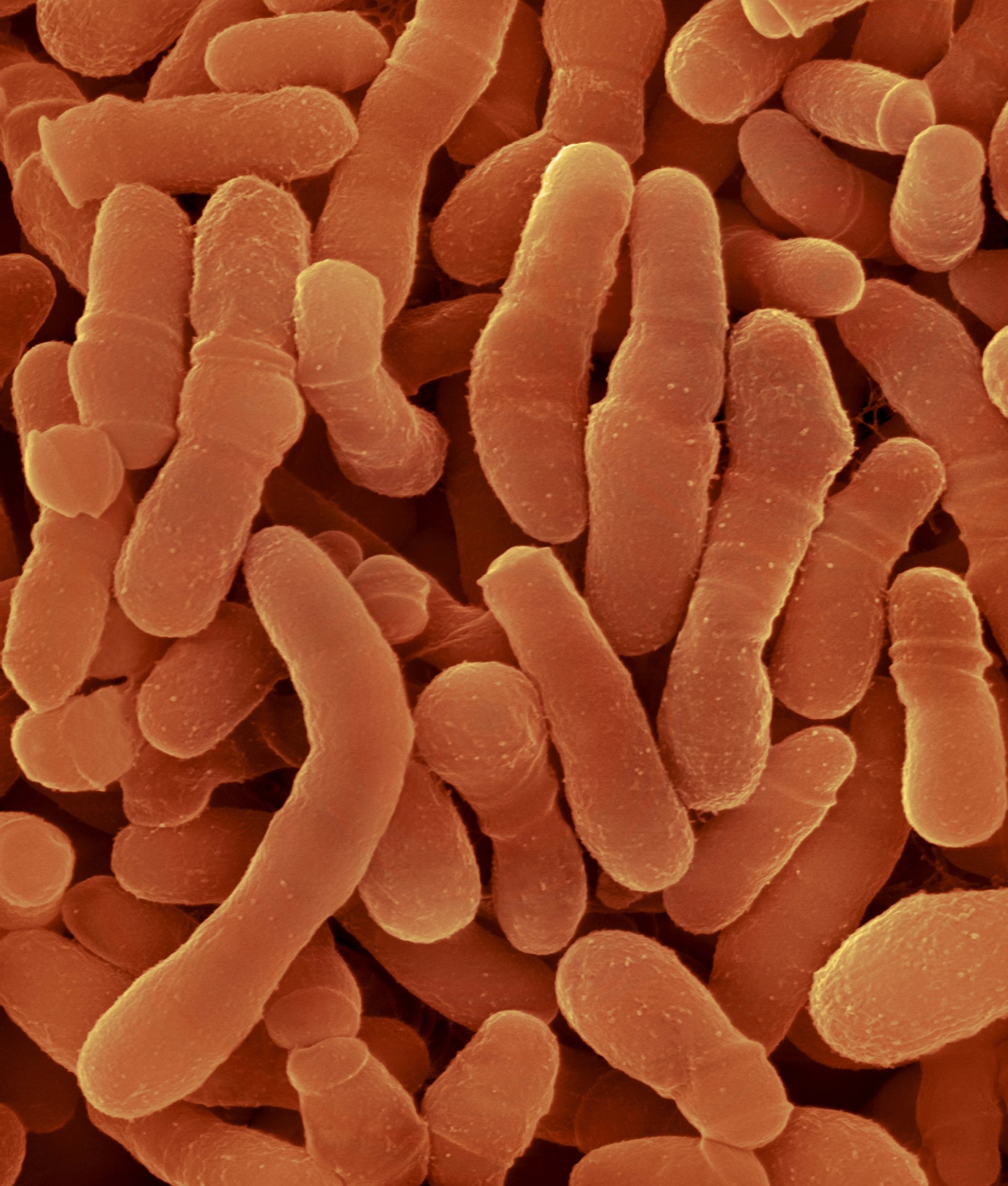
In 2021, ARTIS Micropia Professor Remco Kort and his students studied the faeces of the western lowland gorilla. They did this by comparing the composition of the microbiome of the ARTIS gorillas with that of their wild counterparts – and also with that of humans. They were assisted by Walter Pirovano and Mark Bessem of the company BaseClear, experts in the field of microbial DNA analyses.This research revealed interesting correspondences. In both primate species, a change in lifestyle led to adjustments in their microbiome. So what does this mean?
Big changes
Millions of years ago, we lived as hunter-gatherers in small groups, ranging over a large area. Only in the last 10,000 years did this began to change, triggered by the invention of agriculture. The most drastic change in our lifestyle has resulted from industrialisation over the last 200 years. This latter change has strongly reduced our contact with nature, and hence also with the microbial life that forms part of it – because of things like clean drinking water, the use of antibiotics, disinfectants and an urban environment with many people living in a limited area. Our high-fibre food has been replaced by easily digestible sugars.
The price of progress
This modern, low-microbe lifestyle has brought us many benefits. Our life expectancy is significantly higher than that of hunter-gatherers, and we’ve been able to combat most infectious diseases. But now the question arises whether we also pay a price for this progress: the evidence is mounting that this is indeed the case. Reduced contact with micro-organisms, especially early in life, leads to an immune system that is poorly trained and overreacts to relatively harmless stimuli.
Our microbiome, the microbial ecosystem in and on our body, contains billions of micro-organisms with which we live in symbiosis. And this discovery has led to an important insight. Our immune system not only needs to neutralise harmful micro-organisms – it also needs to tolerate harmless ones. Reduced contact with micro-organisms leads to a higher risk of chronic inflammatory diseases, which often occur in industrialised countries.
Lost microbes
It is exciting to find out how the microbiome of the hunter-gatherers differs from ours. Analyses show that the gut microbiota of hunter-gatherers in Tanzania are more diverse, containing fewer antibiotic-resistant species and many more bacteria species that are able to break down fibres. Generally speaking, the greater the diversity of the gut microbiota, the more resilient the organism to disease.
A comparable picture of high diversity was obtained in the analysis of the microbiota in the fossilised faeces of population groups from the US and Mexico a thousand years ago. So, over the course of time our gut microbiome has become depleted. It is clear that reduced contact with nature and microbes early in life raises the risk of inflammatory diseases. However, it is far from clear whether enriching our microbiome with these lost species will also bring benefits for our health.
Understanding humans
‘Each time I visit Artis, I understand humans better than before.’ This quote by the poet Jan Hendrik Leopold, to be seen at the back of the De Volharding building in ARTIS, also applies to the microbiome really well. By studying the gut microbiome of primates, we can also learn more about our own health and evolution. Last year at ARTIS, we examined faeces samples from the western lowland gorillas, and we also compared these with samples from their wild counterparts.
This study revealed that their microbial diversity is lower and that some microbe species have disappeared. To give one example, bacteria of the genus Olsenella, specialists in digesting fibres, were found less often and in smaller quantities in ARTIS gorillas. We see precisely the same trend when we compare the microbiome of hunter-gatherers with that of modern humans. So the studies show that a long-term stay in a zoo affects the gut microbiome of non-human primates in a way comparable to how city life affects humans.

Silverback Akili prefers willow branches.
Enriching the gorilla diet
So how can we put these insights to use? It is assumed that intestinal problems occur more often among primates in zoos than among primates in the wild. The gorillas in ARTIS, too, suffer from loose stools with some regularity. It may be that the low fibre content of their food contributes to these symptoms. Moreover, this leads to impoverishment of the gut microbiota, because gut bacteria with the specific enzymes for breaking down complex carbohydrates (fibres) are no longer necessary.
This is why we at ARTIS carried out an intervention in which we give the gorillas extra high-fibre branches to eat. When we offer the branches together with other food, Akili – the silverback and leader of the group – prefers the branches. He climbs to the top of the tree in the middle of his outdoor enclosure with them, where he strips them and nibbles the bark. This fibre-rich food is hard to break down and will probably lead to an enrichment of Akili’s gut microbiota, due to a strong increase in specific bacteria (such as those of the genus Olsenella), which have extra enzymes to digest fibres.
Gorilla faeces in close-up
In the following phase of the research, we collected Akili’s faeces in order to isolate the DNA and RNA it contains. The DNA gives us a picture of the gut microbiota, as well as of the food (plants) in the faeces. The RNA gives us a specific picture of the activity of these organisms, because this encodes the information about which genes are active. This activity changes fast, so the gorilla keepers went to great lengths to collect and freeze Akili’s faeces as quickly as possible; this allows for a more representative picture of what is happening in his intestines.
The research is still at an early stage, but we can already give a sneak preview. in addition to the activity of familiar (lactic acid) bacteria, we have also found active fungi such as Piromyces. Since this fungus plays a key role in the digestive tract of other herbivores (plant eaters), this is an interesting result. That is because Piromyces provides hydrogen to methane-producing archaea bacteria in their hosts’ intestines, and moreover helps to digest plant cell-wall material. This material is converted into glucose and other simple sugars, making it available to the host animal. By comparing the gorilla microbiome before and after eating extra willow branches, we hope to learn how fungi and bacteria in the gut collaborate in the digestive process and so contribute to Akili’s health. By the way, increasing the level of high-fibre food in one’s diet is recommended not only for gorillas, but for people as well.
Text Remco Kort
Picture Ronald van Weeren